
E-cigarette exposures, respiratory tract infections, and impaired innate immunity: a narrative review
Introduction
Electronic cigarettes (e-cigarettes) are devices that allow for aerosolization of liquid (e-liquid). Since their introduction, popularity of e-cigarettes has increased significantly with as many as 19.6% of United States high school students reporting current use in 2020 (1,2). Initially touted as a smoking cessation aide, evidence from multiple meta-analyses remain limited and low-grade (3-5), providing general perception to the public that e-cigarettes use is safe and with limited harm.
Rise in popularity of e-cigarettes has promoted continued evolution of the e-cigarette components including the e-liquid and electronic delivery device (e-device). E-liquids consist of three components: a humectant such as propylene glycol (PG) or vegetable glycerin (VG); a drug such as nicotine, tetrahydrocannabinol (THC), or cannabidiol (CBD); and a flavoring chemical to help with tolerance of the aerosol inhalation. Components of e-devices include an atomizer with wick and heating element, battery, and microprocessor. With product evolution, users have learned to modify these components in order to customize use, introducing further complexity to the content and properties of the generated aerosol (6).
As the number of e-cigarette users has increased, so have reports of health related complications. Prior to 2019, published case reports and national reporting was limited, however, E-cigarette, or Vaping, Product Use Associated Lung Injury (EVALI) (7) increased the identification and national reporting of e-cigarette related health complications within the United States. After thousands of hospitalizations and hundreds of deaths (8), national reporting of EVALI cases has slowed. However, concerns remain with regards to the underlying etiology of EVALI as well as other direct and indirect effects of vaping on lung health.
Respiratory infections are one of the leading causes of both pediatric and adult morbidity and mortality in the United States (9). Influenza and respiratory syncytial virus (RSV) are two of the most common respiratory viral infections (RVIs) across all ages, with an estimated 38 million US cases of influenza during the 2019–2020 influenza season (10) and 2.1 million outpatient visits for RSV annually (11). As of January 2021, the pandemic of the novel virus, severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) is ongoing, with greater than 80 million cases of COVID-19 globally thus far (12). Pediatric cases of RVIs are a significant public health problem; pediatric cases make up more than 75% of all RSV cases and one third (32.7%) of all influenza cases (13). Incidence of COVID-19 among pediatric and young adult populations has been increasing dramatically since May 2020 (14). Overall, RVIs are among the top ten leading causes of death in the US in every age group (15). While bacterial respiratory infections are more common in adults than children, respiratory infections secondary to bacterial pneumonia still represent a significant healthcare burden in the US with Streptococcus pneumoniae being the primary cause of bacterial pneumonia in both children and adults (16). Despite the introduction of a vaccine, Haemophilus influenzae infections continue to represent a significant portion of lower respiratory tract infections within the US as well (17).
The primary purpose of this editorial is to review the current evidence of e-cigarette use and their association with respiratory tract infections. The primary emphasis of most preclinical research is the effect of e-cigarette exposure on the innate immune system, the main initial barrier to respiratory infections. Such information may provide guidance to healthcare providers, policy advocates and researchers for making informed decisions regarding associated respiratory health risks and e-cigarette use. The following article is in accordance with the Narrative Review reporting checklist by Pediatric Medicine (available at http://dx.doi.org/10.21037/pm-20-97).
Methods
Literature searches were conducted in PubMed, CINAHL and MEDLINE for articles published after the year 2008 using the following keywords: “electronic cigarette”, “electronic nicotine delivery system”, “ENDS”, “e-cig”, “e-cigarette”, “e-liquid”, “vape”, “vaping”. Original articles on e-cigarettes were selected if they were in English or available in English and were published in a peer-reviewed journal. The titles and abstracts were then screened for the following criteria: (I) pertaining to respiratory tract infections (either, bacterial, viral or fungal infections associated with the respiratory tract) or pertaining to innate immune defense against respiratory tract infections and (II) having exposure to e-cigarette vapor (ECV), aerosol, or e-cigarette extract. Those studies that did not involve both criteria and pertain to original research on e-cigarettes were not included. Previously published manuscripts identified by the authors on the topic were also included in the final list of references for review.
E-cigarettes devices and e-liquid overview
Electronic nicotine delivery systems (ENDS), or e-cigarette devices (e-device), are devices that aerosolize chemicals (Figure 1). ‘Vaping’ is the act of inhaling heated and aerosolized chemicals. ENDS devices consist of a battery, atomizer, microprocessor, pneumatic sensor and container or cartridge for the solvent mixture, or “e-liquid”. When a user inhales, the pneumatic sensor activates the microprocessor that in turn instructs the battery to send energy to the heating coil inside the atomizer. The heat from the coil then aerosolizes the e-liquid contents, and the user is able to inhale the compounds within the e-liquid. E-cigarette devices can be modified by varying the voltage, requiring changes to the battery and associated heating element.
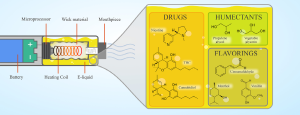
E-liquid consists of three components: drug, flavoring chemical, and humectant. Three of the most common drugs inhaled using e-devices are nicotine, THC, and CBD. Flavorings are chemicals added to e-liquid to make the inhaled aerosol more palatable, with variations in the smell, flavor, and texture. Examples of flavorings include aliphatic aldehydes, most common in fruity flavors, and aromatic aldehydes, often seen in spicy flavors (18). Finally, the humectant is the chemical that binds the drug and flavorings while providing moisture. The two most common humectants are PG and VG (19). The recent E-cigarette, or Vaping, Product Associated Lung Injury (EVALI) pandemic highlights the importance of additional chemicals that may be added to e-cigarette cartridges to dilute the primary inhaled drug. Vitamin E acetate (VEA) is the primary chemical identified (20) and strongly associated with most EVALI cases (8). VEA is added to many cartridges to dilute the concentration of THC in order to maximize a seller’s profit while providing similar liquid viscosity. Direct exposure to VEA via inhalation exposure in mice has been shown to cause acute lung injury (21). Thus, reducing or eliminating VEA in e-liquids most likely contributed to the reduced incidence of EVALI cases requiring hospitalization since 2019. However, other viscous chemicals with similar properties and composition to that of VEA may have similar effects on the respiratory tract. There is also evidence that some e-liquids contain other impurities and toxic derivatives not intended by the manufacturer, such as heavy metals or heat transformed byproducts such as ketene (22) or acrolein (23). The purpose of this introduction is not to be all encompassing on e-cigarettes or ENDS product design [as has been done previously (24-26)], but to provide a brief introduction into the associated chemical and heating elements that may contribute to e-cigarette associated respiratory pathology.
Innate immunity of the lung and the effect of e-cigarette exposure
The respiratory tract has multiple layers of defenses that protect the lung from infection. These are broadly grouped into the innate and adaptive immunity (27). The adaptive immunity is a highly complex and coordinate section of the immune system, including both cellular and humoral sub-components and eloquently reviewed previously (28,29). The primary focus of this review is on how e-cigarette inhalation exposure modifies the lung’s innate immune defense mechanisms (summarized in Figure 2). The innate immune system in the lung is the first line of defense for fighting off invading pathogens. Components of the innate immunity include the airway surface liquid (ASL), the respiratory epithelium including its cilia, humoral and cellular responses. Humoral innate immunity includes soluble factors such as LPS binding protein (LBP), collectins, opsonins, defensins and various components of the complement system. Many of these soluble factors also assist or prime the adaptive immune system response to invading pathogens. Conversely, the cellular components of the lung’s innate immunity include the ASL, airway epithelial cells, macrophages, and neutrophils. The following paragraphs highlight the function of each component as well as the effect of e-cigarette exposure on each component’s function (also highlighted in Table 1).
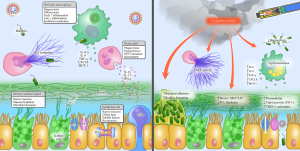
Table 1
| First author (year) journal | Primary cell | Model | Summary of primary results |
|---|---|---|---|
| In vivo experiments | |||
| Bagale K et al. [2020] Appl Environ Microbiol (30) | Epithelium | S. pneumoniae in C57BL/6 mice | Exposed S. pneumoniae to EVE, then inoculated with bacteria |
| No difference in lavage cytokine response following S. pneumonia inoculation | |||
| Increased bacterial biofilm formation when mice are exposed to EVE with nicotine | |||
| Corriden R et al. [2020] Am J Physiol Cell Physiol (31) | Neutrophils | P. aeruginosa in C57BL/6 or CD-1 mice | Direct exposure of mice to 60 minutes daily of EV for 1 month |
| Increased P. aeruginosa burden in peritoneum with e-cig exposed versus air controls | |||
| Reduced neutrophil migration with e-cig exposed versus air controls | |||
| Hwang JH et al. [2016] J Mol Med (Berl) (32) | Epithelium | S. aureus in CD-1 mice | Direct exposure of mice to 60 minutes daily of EV for 1 month |
| Increased biofilm formation, increased adherence and invasion of S. aureus | |||
| Up-regulation of S. aureus-specific virulent genes and increased virulence after S. aureus exposed to e-cigarette extract | |||
| Madison MC et al. [2019] J Clin Invest (33) | Macrophages | Influenza A (H3N2) in C57BL/6 mice | Direct exposure to EV for 24 total minutes daily for 1 month |
| Increased weight loss and mortality after H3N2 inoculation | |||
| Increased lung tissue inflammation and architectural distortion | |||
| Increased IFN-γ (EV + nicotine) and increased TNF-α (EV-only) | |||
| Sussan TE et al. [2015] PLoS One (34) | Macrophages | S. pneumoniae or influenza A (H1N1) in C57BL/6 mice | Direct exposure to EV for 90 minutes daily for 2 weeks |
| Increased extracellular S. pneumoniae burden from lavage cultures after inoculation | |||
| Increased H1N1 viral titers from lavage after exposure | |||
| Increased illness recovery, mortality and weight loss after H1N1 inoculation | |||
| Increase in IL-6, MCP-1 and decrease in IL-17, IFN-γ in H1N1 inoculated lavage samples | |||
| In vitro experiments | |||
| Crotty Alexander LE et al. [2018] Am J Physiol Regul Integr Comp Physiol (35) | Epithelium | P. aeruginosa in human airway cells | Increase in trans-epithelial permeability during P. aeruginosa infection |
| Increased IL-8 levels during P. aeruginosa infection | |||
| Gómez AC et al. [2020] PLoS One (36) | Macrophages | M. tuberculosis in THP-1 cells | No effect on cell viability |
| Decreased phagocytosis of M. tuberculosis after EVE exposure, independent of nicotine | |||
| Hwang JH et al. [2016] J Mol Med (Berl) (32) | Epithelium | S. aureus in primary human airway cells | Increased cell death (>50%) after direct exposure to EVE |
| Increased cell death after direct application of 2 mg/mL of nicotine alone | |||
| Increased cell death (40-99%) after exposure to 4 out of 6 e-liquid brands tested | |||
| Corriden R et al. [2020] Am J Physiol Cell Physiol (31) | Epithelium | S. pneumonia in nasal and bronchial cells | Increased PAFR expression |
| Increased pneumococcal adhesion to nasal and bronchial ECs | |||
| Scott A et al. [2018] Thorax (37) | Macrophages | E. coli in THP-1 cells | Decreased phagocytosis following e-cig exposure with and without nicotine |
| Ween MP et al. [2017] Phys Rep (38) | Macrophages | Non-typeable H. influenza (NTHi) in THP-1 cells | Reduced phagocytosis of NTHi with flavored e-liquids |
| Reduced SR-A1 receptor expression following e-liquid exposure | |||
| Increased IL-8 and reduced TNF-α, IL-6, IL-1b with 2 of the 3 flavors | |||
| Wu Q et al. [2014] PLoS One (39) | Epithelium | HRV-16 in primary human bronchial cells | Reduced SPLUNC1 mRNA expression levels in ECs |
| Increased IL-6 production independent of nicotine |
†, peer-reviewed literature included within this table are those studies that met both criteria: (I) exposure to e-cigarette aerosol or aerosol extract in a small rodent model or lung-specific cell type and (II) a secondary inoculation with a respiratory pathogen. EVE, e-cigarette vapor extract; EV, e-cigarette vapor; ECs, epithelial cells.
ASL, surfactant proteins (SP) and mucin production
The ASL consists of a thin layer of fluid (40) covering the luminal surface of the conducting airways. It is composed of water, electrolytes and mucins separated into two layers of different consistency. The lower layer, known as the periciliary liquid layer (PCL), is a poly-anionic low friction gel layer (41) thought to promote efficient ciliary beating. The upper layer consists of high molecular weight mucins, such as MUC5B and MUC5AC, resulting in a tangled, unrestrained gel. ASL hydrates the airways and allows for expulsion of trapped toxins and microbes via mucous clearance (42). In addition the ASL acts as a direct physical barrier to microbial invasion and regulates airway acidity (pH) which is known to facilitate phagocytosis and efferocytosis in response to bacterial colonization (43). Failure to maintain a hydrated, intact ASL results in thicker mucus, leading to decreased mucus clearance, recurrent infections and ultimately contributing substantially to the development of bronchiectasis (44). In contrast to the gel-like ASL within the conducting airways, the alveoli are lined with a thin watery liquid known as alveolar sub-phase fluid (AVSF) and a layer of pulmonary surfactant film. Pulmonary surfactant consists of phospholipids and several proteins known as SP some of which (more specifically, SP-A and SP-D) play an important role in antimicrobial defenses. SP-A and SP-D inhibit pathogens directly and recruit alveolar macrophages (AM) via chemotaxis.
ECV has a negative effect on normal ASL function. Healthy human bronchial epithelial cells (HBEC) differentiated at air-liquid interface (ALI) showed a reduction in ASL volume and an increase in ASL viscosity when exposed to ECV plus nicotine compared to ECV or control exposure alone (45). This effect was reproduced with nicotine solution alone, independent of ECV, with increased ASL viscosity and decreased ASL volume that was dose dependent. To further delineate the underlying mechanisms contributing to this effect, the researchers assessed transient receptor potential ankyrin 1 (TRPA1) activity, a known irritant channel and target of nicotine and e-cigarette flavors such as cinnamaldehyde (‘cinnamon flavor’). When HBEC (the primary cells that produce ASL) were exposed to a selective TRPA1 agonist, ASL volume was reduced with increased ASL viscosity increased. Conversely, HBECs in the presence of a TRPA1 antagonist prevented the reduction in ASL volume and increase in viscosity when exposed to nicotine. Additionally, when sheep tracheas were exposure to ECV, tracheal mucous velocity was reduced compared to control, suggestive of impaired mucociliary clearance. Hence, ECV exposure with nicotine significant reduced ASL volume and increased viscosity through activation of TRPA1.
A second target of ASL is cystic fibrosis transmembrane conductance regulator (CFTR). CFTR is an ion channel protein that conducts chloride and sodium across the airway epithelial membrane. When CFTR is dysfunctional, less sodium is exchanged, which in turn dehydrates ASL making viscous mucus. A one-hour exposure of e-cig aerosol to HBECs differentiated at ALI significantly inhibited CFTR function compared to air-exposed controls (46). Interestingly, when the e-device was replaced by a nebulizer, no significant inhibition of CFTR function occurred, suggestive that temperature or heat transformation may have a significant impact on CFTR function after e-cig aerosol exposure. Mass spectrometry confirmed the presence of acrolein in e-cigarette aerosol using the e-device, a reactive aldehyde commonly produced in cigarette combustion and a well-known inhibitor of CFTR. The amount of acrolein produced was proportional to the power generated by the e-device. These studies suggest that CFTR function and its subsequent contribution to ASL hydration is partially inhibited by the toxic derivative, acrolein. Less studied are the effects of various flavorings added to e-liquids on CFTR function. In a study assessing the safety of seven common flavoring chemicals on HBECs, vanillin and 2,5-dimethylpyrazine (‘chocolate’) were two chemicals found to alter epithelial cellular signaling profiles. In subsequent experiments using mouse tracheal cells, chocolate flavoring induced a rapid activation of chloride current through CFTR via the protein kinase A pathway (47).
A separate research team reported that the ASL from healthy human donor was not affected by e-cigarette aerosol with nicotine or nicotine alone (48). Ciliary beat frequency (CBF), as measured by a microscope with a slow-motion camera, was likewise unaffected. The same team also analyzed the epithelial sodium channel (ENaC) and CFTR function, finding no effect of e-cigarette aerosol or nicotine. One explanation for the differences from prior studies is that this study used a short (5 second) puffing duration and collected the aerosol through a filter before diluting and applying to HBECs. This is in contrast to the prior studies, which use direct exposure to e-cigarette aerosol for a longer puff duration and for a greater number of total puffs.
How e-cigarettes affect the individual components of the ASL and AVSF are also in the early stage of research. A lung surfactant substitute derived from calf lungs with similar biophysical properties to human surfactant and exposed to aerosolized e-liquid constitutions at various concentrations of PG and VG showed no significant difference in physical properties (surface tension, elasticity) of the surfactant substitute (49). There was a decrease in surface tension and elasticity but only at PG/VG concentrations of >200× of what the researchers predicted would be present in a normal puff. A similar study also did not show any changes to surface pressure of a surfactant substitute compound after exposure to e-vapor (50). Conversely, VEA, the chemical found in e-liquids for a majority of EVALI cases (51), increased surfactant compressibility when added to a lung surfactant substitute (52). Compressibility is the ability of liquid to deform under pressure. Hence, VEA may compress the body’s innate surfactant, predisposing to alveolar collapse on exhalation, and contributing to subsequent respiratory failure, as was seen with EVALI.
The evidence base detailing the effects of e-cigarettes on mucins is also growing. Induced sputum samples from e-cigarette users were compared to those from tobacco smokers and never-smokers. A significant increase in concentration of MUC5AC, and thus in the MUC5AC/MUC5B ratio, in sputum of e-cigarette users compared to non-smokers, with similar results seen in tobacco smoking (37). Elevated MUC5AC/MUC5B ratio has previously been shown to correlate with mucus stasis and obstruction in asthmatics (53). A study of HBECs cultured at ALI similarly showed an increase of MUC5AC and IL-6 when exposed to nicotine-free e-vapor (54). Prior research has shown that IL-6 increases mucin production (55), so the study authors administered an anti-IL-6 which normalized MUC5AC levels suggesting that ECV increases MUC5AC via IL-6.
E-cigarettes have several effects on the barriers of the innate respiratory immune system. These effects may greatly vary by the composition of the base e-liquid, the presence of nicotine as well as the addition of any of the myriad of flavoring chemicals or other constituents added. Literature reviewed above suggests e-cigarette exposures reduces the volume of ASL possibly mediated via activation of TRPA1 and inhibition of CFTR. Inversely, a ‘chocolate’ flavor additive has been shown to acutely activate CFTR. At the same time e-vapor increases the amount of MUC5AC within the mucin gel via an increase in IL-6. Collectively, these results of increased MUC5AC and CFTR dysregulation may lead to increasing mucous plugging, increasing the risk of bronchitis and lower respiratory tract infections. Future therapeutic targets that function as mucolytics, anti-inflammatories, and/or anti-oxidants, such as N-acetylcysteine (NAC) may provide future benefit for those with increased and more viscous mucus secondary to e-cigarette exposure.
Airway epithelial cells
Lung epithelial cells play multiple roles in the lung’s innate immune response (27). Lung epithelial cells provide a physical barrier to the external environment preventing microbial invasion. The airway epithelium consists of greater than 40 different subpopulations of epithelial cells. Some of the most abundant of these subpopulations include goblet, club and ciliated cells (56-58). Goblet cells secrete various mucins into the airway lumen to provide an additional layer of barrier protection to the external environment. Club cells contain also secrete a variety of products to protect the airway including uteroglobin and other various glycosaminoglycans (56). In addition to their exocrine function, club cells contain a high concentration of cytochrome P450 enzymes for detoxifying harmful inhaled substances. One of the most abundant airway epithelial cell types ciliated cells transport mucous proximally by continuous and coordinated beating of apically oriented cilia on the airway lumen, clearing inhaled debris and microbes. Primary cilia on epithelial cells can also provide chemosensation, thermosensation and mechanosensation for sensing the external airway environment (59). The airway epithelium is endowed with many additional antimicrobial functions, eloquently highlighted by prior review articles (60-62). Microbial invasion of the airway epithelium can also be the sentinel event to other signaling cascade triggering activation of the innate immune response (63). Dysfunction of any one of these inherent elements of the airway epithelium can result in increased susceptibility to respiratory infections.
Previous literature evaluating the effect of e-cigarette aerosol exposures on the lung epithelium have focused primarily on the proximal airway epithelium using co-exposures to certain viruses or bacteria. Normal human bronchial epithelial (NHBE) cells grown at ALI and then exposed to e-cigarette aerosols containing PG/VG + nicotine for 15 minutes daily for 2 to 5 consecutive days resulted in increased permeability as measured by fluorescein isothiocyanate (FITC)-dextran penetrance through a porous transwell membrane (35). The expression of the tight junction protein zona occludens (ZO1) decreased in e-cigarette-exposed cultures compared to air-exposed controls, suggestive of reduced tight junction adhesion contributing to increased permeability. When the bronchial epithelial cells were infected subsequently with Pseudomonas aeruginosa, a gram-negative bacteria pathogenic in susceptible populations with underlying lung disease such as cystic fibrosis, IL-8 expression in e-cigarette-exposed NHBE’s increased significantly compared to air controls infected with P. aeruginosa. IL-8 is a chemoattractant cytokine specific for attracting and activating neutrophils. Hence, these in vitro in human airway cultures suggest prior e-cigarette aerosol exposure to augment a neutrophilic chemotactic response to certain pathogenic bacteria such as P. aeruginosa.
Streptococcus pneumoniae is the most common bacteria to cause community acquired pneumonia in both children and adults (64). S. pneumoniae has been shown to enter human airway epithelial cells by adhering to the platelet-activating factor receptor (PAFR), which is then internalized by the cell (65). Whether this adhesive ability is affected by e-cigarettes has been examined both in vitro using cultured human airway epithelial cells as well as in vivo using a mouse pneumococcal colonization model (31). For in vitro experiments, human nasal epithelial cells were obtained from e-cigarette users before and one hour after vaping, and compared to healthy non-vaping controls. Baseline expression of PAFR was similar between e-cigarette users and healthy controls, however, PAFR expression significantly increased one hour after e-cigarette use. When the alveolar type II epithelial cell line A549 were exposed to condensates from ECV and were subsequently inoculated with the S. pneumoniae strain D39, pneumococcal adhesion and internalization increased significantly in ECV-exposed cells compared to non-exposed controls. Furthermore, the PAFR blocker (CV3988) applied to human primary bronchial epithelial cells attenuated pneumococcal adhesion stimulated by ECV. Using a mouse model of pneumococcal colonization, female CD1 mice were dosed with ECV for 4 days, followed by exposure to 1×105 colony-forming units (CFU’s) of S. pneumoniae. Mice exposed to nicotine-free ECV alone failed to increase nasopharyngeal S. pneumoniae colonization or PAFR expression compared to air controls, while those exposed to nicotine-rich ECV showed both increased colonization and PAFR expression. In summary, Corriden et al. (31) conclude that ECV exposure increases S. pneumoniae adhesion to lung epithelial cells and asymptomatic S. pneumoniae colonization in mice partially through increased PAFR expression. To evaluate whether ECV exposure affected S. pneumoniae virulence, Bagale et al. (30) inoculated C57BL6 mice with the S. pneumoniae strain TIGR4 after exposing the bacteria to ECV. No significant difference in bacterial burden or respiratory cytokine profiles from mouse respiratory lavage was identified between ECV-exposed and unexposed bacteria. However, there was a moderate increase in biofilm formation from S. pneumoniae exposed to ECV with nicotine. As bacterial colonization is thought to play a key role preceding certain respiratory infections (66), these preclinical studies suggest e-cigarette exposure to airway epithelial cells, and not to S. pneumonia bacteria alone, may increase susceptibility to S. pneumonia colonization and subsequent pulmonary exacerbations or acute pneumonia.
Staphylococcus aureus (S. aureus) is a second common bacteria seen with severe pneumonia and septicemia. At least twenty percent of the human population is colonized with S. aureus in the anterior nares, which is thought to be an important step in the development of clinical infections (67). Hwang et al. evaluated whether e-cigarettes affect S. aureus virulence, colonization and respiratory epithelial cell immune response (32). In vitro significant cytotoxicity occurred in human keratinocytes exposed to ECV extract (EVE) or exposed directly to EV independent of nicotine concentration. Following inoculation with methicillin-resistant S. aureus (MRSA), epithelial cells exposed to EV had greater MRSA bacterial burden compared to non-EV-exposed epithelial cells. When MRSA itself was exposed to EV, several of its virulence factors were amplified including its biofilm formation, hydrophobicity and adherence to epithelial cells as well as increased resistance to LL-37 (anti-microbial peptide released by airway epithelial cells). In vivo, mice that inhaled ECVs for 1 hour per day for 1 month demonstrated no change in bronchoalveolar lavage cell counts compared to air-exposed controls. However, mice infected with EV-exposed MRSA worse mortality compared to those infected with non-exposed MRSA. These studies suggest e-cigarette exposure to MRSA may significantly increase its virulence through not one but multiple mechanisms including increased biofilm formation, epithelial adhesion and increased bacterial replication. Assuming a true bacterial infection with MRSA following e-cigarette exposure, antibiotics that target these mechanisms that are specific to MRSA, such as vancomycin or linezolid, may reduce its virulence.
Human rhinovirus (HRV) is the most common pathogen of acute upper respiratory tract infections well known to provoke acute lower respiratory exacerbations in susceptible populations with asthma and chronic obstructive pulmonary disease (COPD) (68). To assess whether e-cigarette exposure increased susceptibility to HRV infection, Wu et al. evaluated whether acute e-cigarette liquid exposure containing various concentrations of nicotine in culture with human trachea and bronchi epithelial (hTBE) alters airway epithelial function against HRV infection (39). Interleukin-6 (IL-6), a potent pro-inflammatory cytokine, expression was measured in epithelial culture supernatants at baseline, 6 and 24 hours after e-cigarette liquid exposure. hTBE’s treated with e-liquid compared to media alone, and independent of nicotine, increased IL-6 secretion in epithelial supernatants. Following inoculation with HRV serotype 16 (HRV-16), e-liquid-treated hTBE’s demonstrated higher viral loads at 6 and 24 hours, again independent of nicotine, compared to hTBE’s with media and HRV-16 alone. SPLUNC1 mRNA expression, was concurrently significantly reduced in hTBE’s treated with e-liquid compared to cultures without e-liquid. SPLUNC1 is a gene commonly expressed in airway epithelium whose associated protein is closely associated with multiple antimicrobial properties (69). SPUNC1 knockout (KO) C57BL6 mice demonstrate elevated levels of HRV-1B gene expression compared to wild-type C57BL6 control mice following HRV-1B intranasal inoculation. Collectively, these results suggest e-cigarette exposure in airway epithelial cells may promote HRV infection through pro-inflammatory signaling cascades and reduced SPUNC1 gene expression.
The previous studies provide early preclinical findings to suggest that e-cigarette exposure to airway epithelial cells promotes bacterial and viral virulence following direct inoculation. Implicated pathogens commonly associated with the respiratory tract and identified as pathogenic in preclinical models include bacteria P. aeruginosa, S. pneumoniae, S. aureus as well as HRV. The exact mechanism of how e-cigarette exposure to airway epithelial cells contributes to increased virulence remains poorly understood, however, these early studies suggest injury to the airway epithelium being a sentinel event in the pro-inflammatory signaling cascade promoting bacterial or viral infections.
Lung macrophages
Lung macrophages can be classified as AM and interstitial macrophages (IM) based on their underlying origins and histologic features (70). Lung AM’s are self-renewing whereas IM’s are typically derived from blood monocytes (70). AM’s play an essential role in lung lipid homeostasis and lung repair after injury in addition to their central pro- and anti-inflammatory responses during pulmonary infections (71,72). In the initial phase of microbial invasion, AMs ingest and phagocytose pathogens to be destroyed by reactive oxygen species (ROS) and acidic phagosomes (73). AMs then initiate an inflammatory response by releasing pro-inflammatory cytokines such as interleukin (IL)-1, IL-6, and tumor necrosis factor (TNF)-α that promote the recruitment of additional inflammatory cells to the lungs. As this response accelerates, both AM’s and IM’s can dampen further inflammatory responses by ingesting dying cells, a process known as efferocytosis, while releasing anti-inflammatory cytokines such as IL-10. Lung macrophages can also catabolize damaged surfactant (74) to maintain an intact ALI at the epithelial layer.
As lung macrophages are a vital component of the respiratory innate immunity, Sussan et al. (34) evaluated the effects of e-cigarette vapor (EV) exposure on lung macrophage function. Lung macrophages harvested from mice exposed to EV had a 58% increase in number of lung macrophages with a concurrent decrease in IL-6 expression compared to air control lung macrophages. A sub-population of mice exposed to EV were subsequently inoculated intranasally with S. pneumoniae after the last dose of EV. Mice exposed to EV had a significant increase in pulmonary S. pneumoniae colony formation from lung lavage cultures. Macrophages from EV-exposed mice inoculated with S. pneumoniae also demonstrated a significant increase in extracellular bacterial burden with a simultaneously decrease in intra cellular bacterial burden compared to controls, strongly suggestive of poor macrophage phagocytosis. In additional studies, EV-exposed mice inoculated with or mouse-adapted influenza A (H1N1) showed significantly elevated H1N1 viral titers in the lavage, delayed illness recovery time, increased mortality (20% vs. 0%), and worsening of percent weight loss compared to H1N1 inoculated mice without prior EV exposure. Lavage cytokine analyses at day 8 after H1N1 inoculation failed to demonstrate a statistically significant difference in EV-exposed mice compared to controls, however, the pro-inflammatory cytokines of IL-6 and MCP-1 trended towards an increase while IL-17 and IFN-γ trended lower in mice exposed to EV-exposed mice compared to non-EV-exposed controls.
Similar results of increased pro-inflammatory signaling have been shown using cultures of human-derived lung macrophages. Lung macrophages isolated from never smokers exposed to ECV condensate (ECVC) containing nicotine for 24 hours in vitro showed significantly reduced viability with increased apoptosis and necrosis compared to controls (37). Nicotine-free ECVC increased apoptosis alone, suggesting increased cytotoxicity with the addition of nicotine (37). ECVC-exposed macrophages also showed a significant increase in ROS production independent of nicotine concentration. Increased production of IL-6, TNF-alpha, CXCL8, MCP-1 and MMP-9 were seen in ECVC-exposed macrophage cultures compared to controls. Finally, ECVC-exposed macrophages reduced phagocytosis when incubated with E. coli bio-particles compared to non-exposed control macrophages. In a separate study, THP-1-derived macrophages exposed to ECVC and then challenged with Mycobacterium tuberculosis for 6 days demonstrated reduced phagocytosis independent of nicotine (36). Collectively, these studies of preclinical rodent models and in vitro human macrophage challenge models suggest e-cigarette exposure may significantly impair lung macrophage function, most commonly through impaired phagocytosis of invading pathogens.
Additional chemicals used for e-cigarette flavorings may also impair macrophage function. Ween et al. (38) showed a significant reduction in macrophage phagocytosis of non-typeable Haemophilus influenza (NTHi) following exposure to three different e-liquid flavors. The expression of scavenger receptor A1 (SR-A1), a phagocytosis receptor expressed on macrophages to recognize phagocytic targets, decreased significantly with all three ‘apple’ flavors compared to air controls. The cytokine profile of these e-cigarette exposed macrophages showed elevated levels of neutrophil chemoattractant IL-8 and decreased levels of TNF-α, IL-6 and IL-1β. Clapp et al. (75) identified similar results in lung macrophages harvested from non-smoking adults via lavage and exposed to various flavored e-liquids. Macrophages exposed to humectant alone reduced phagocytosis of S. aureus in culture compared to non-exposed controls (75). Exposure to certain flavored liquids, specifically ‘Sini-cide’ and ‘Kola’, significantly reduced phagocytosis of S. aureus beyond exposure to humectant alone. Researchers noted variation in cytokine profiles that was flavoring-dependent with the “Kola” flavor significantly increasing IL-6 while the Sini-cide flavor significantly suppressed IL-6. Hence, e-cigarette flavoring additives adds additional complexity to determining the role of e-cigarette exposure on macrophage function.
Macrophage function in the lung is finely tuned by lipid and surfactant metabolism (76). Madison et al. (33) examined lipid homeostasis in mice exposed to EV for 4 months. Lung macrophages obtained via lavage from mice following EV exposure demonstrated significant expression of lipid inclusions visible by Oil Red O staining that was independent of glycerol expression, suggestive of impaired endogenous lipid metabolism. Further experiments revealed impaired expression of regulatory SP (SP-A and SP-D), unorganized lamellar bodies within type 2 alveolar cells and increased phospholipid pools coating the terminal airway after EV exposure independent of nicotine. Finally, the researchers showed that mice chronically exposed to EV exhibited an increase in mortality and weight loss after inoculation with influenza A (H3N2). Thus, e-cigarette exposure chronically partially impairs lung lipid metabolism contributing to impaired macrophage function and worsening morbidity following flu exposure in mice.
Collectively, these in vitro and in vivo studies suggest e-cigarette exposure may impair macrophage function in the setting of a respiratory tract infection via reduced phagocytosis. A variety of common respiratory pathogens implicated in these preclinical models includes H. influenzae, S. aureus, influenza virus and M. tuberculosis. Concurrently, these studies also demonstrate a change in the inflammatory cytokine profile of lung macrophages after e-cigarette exposure with increased IL-6, IL-8, MCP-1, and TNF-α and reduced IL-1β, IL-17 and IFN-γ. However, cytokine profile and innate immune system modulation are dependent upon the specific e-liquid, method of aerosol exposure and generation, duration, and additional flavoring chemicals added. The unique profiles and immune modulation present in these studies emphasizes the importance of detailed vaping history taking (device, volume, flavors) by clinicians when determining the risk of vaping to a particular patient.
Neutrophils
Macrophages and airway epithelial cells attract additional immune cells to the lung to assist in the anti-microbial response. Chief among these immune cells are neutrophils. Neutrophils can kill microbes directly and recruit other immune cells to an area of infection. While not commonly found abundantly in healthy lung lavage, neutrophils can quickly mobilize from the adjacent pulmonary vasculature when signaled by an inflammatory insult. Once recruited, neutrophils can initiate their bactericidal activity through three distinct mechanisms: (I) phagocytosis exposes microbes to ROS and an acidic environment; (II) degranulation releases cytotoxic granules such as myeloperoxidase (MPO) with direct anti-microbial activity; and (III) neutrophil extracellular traps (NETs) formation trap pathogens in extracellular fibers composed of neutrophil DNA. Neutrophils can also release chemokines CXCL10, CXCR3, and CXCL2 that enhance their own activity and recruit other neutrophils to the area. Finally, neutrophils can also recruit natural killer T-cells via CCL17 and CD8+ antiviral T cells via release of CXCL12 (77).
To examine NET formation and neutrophil activity in electronic cigarette users, sputum and plasma neutrophils were collected from e-cigarette users and NET activity as well as levels of granule proteins were measured (78). A significant increase in neutrophil granule proteins and NET-associated proteins were seen within the sputum of e-cigarette users compared to non-smoking controls. Neutrophils isolated from the plasma of e-cigarette users and exposed to phorbol 12-myristate 13-acetate (PMA), a potent NET agonist, showed more NET activity as measured by neutrophil nucleic acid release over time compared to non-users. In addition, the authors found a significant increase in MUC5AC concentration within the sputum of e-cigarette users compared to non-exposed control subjects. The authors hypothesize that recurrent e-cigarette exposure increases NET formation and neutrophil activation both locally and systemically. NET activation in the systemic circulation could hypothetically lead to more systemic inflammation and subsequent cardiovascular complications such as ischemic stroke or myocardial infarction.
A separate study examined how e-cigarette flavorings affect neutrophil activity (75). Plasma neutrophils from non-smoking, non-vaping adults were exposed to various e-liquids and PMA was then used to trigger NET formation. Neutrophils exposed to humectant alone (PG/VG 1% dilution) had similar NET activity as controls. When “Kola” e-liquid (8.96 uM cinnamaldehyde) was used, there was significantly more chromatin released suggesting expedited NET formation. NET formation was suppressed following exposure to 1% “Hot Cinnamon Candies” e-liquid with cinnamaldehyde. Finally, the authors evaluated the phagocytic function of neutrophils, showing that cinnamaldehyde e-liquid significantly suppressed phagocytosis in a dose-dependent manner. Overall the authors show that cinnamaldehyde, a highly reactive aldehyde common to certain flavored e-liquids, may have a concentration-dependent response on NET formation. Similar to macrophage and epithelial dysregulation, these studies observed flavor dependent changes in NET formation.
Fewer data are available on neutrophil function following e-cigarette exposure in comparison to lung macrophage and epithelial cell populations, however, the three studies highlighted above demonstrate that e-cigarette exposures can result in dysregulation of NET formation and impaired neutrophil activity. Most of the effects of e-cigarette exposure on neutrophils were seen, not only locally within the lung but also, throughout the systemic circulation. These early preclinical studies demonstrate repetitive e-cigarette exposure impairs neutrophilic phagocytosis, placing patients at risk for colonization and clearance of certain respiratory bacteria.
Future clinical context & perspective
In summary, preclinical studies have observed e-cigarette exposure leading to impairment of the lung’s innate immunity. While some of the effects of e-cigarette exposure on the innate immune system are similar to that of combustible tobacco exposure, the emerging literature highlighted above suggests cell-type and pathogen-specific mechanistic differences between e-cigarette and combustible cigarette exposure in dysregulation of innate immunity. Outcomes identified to be cell-type specific include: (I) impaired epithelial barrier function, (II) impaired efferocytosis by lung macrophages, and (III) reduced neutrophilic migration and phagocytosis. Various preclinical studies have used primary human cells in culture as well as in vivo rodent studies inoculated with the common respiratory pathogens of S. pneumoniae, non-typeable H. influenzae, S. aureus, H. influenza, or HRV. Additional endpoints identified that are important to the lung’s innate immunity include dysregulated lipid homeostasis and abnormal cytokine signaling. Collectively, these findings suggest that e-cigarette aerosol exposure significantly impairs the lung’s innate immune response through multiple proposed mechanisms. Targeting one or multiple pathways alone or in combination may provide future therapeutic development.
While significant headway has been made in delineating the effects of e-cigarette inhalation exposure on the respiratory tract, additional work remains to clarify some of the related mechanisms associated with underlying lung disease. To provide greater granularity to these mechanisms, future research is required investigating: (I) chemical constituents of e-cigarette liquids, (II) the aerosolized products of e-cigarette liquids, and (III) individual components as well as mixtures of e-cig liquid components contributing to toxicity. Additional clinical and translational studies are required to validate the identified pathways seen in those preclinical studies highlighted within this review. Clinical evidence is also required to assess whether e-cigarette users, or dual e-cigarette and combustible cigarette users, are at greater risk for the development of respiratory tract infection compared to the general population.
While these identified effects on the innate immunity in the lung have not been studied specifically in the pediatric population, it is plausible that these adverse immune changes, or even systemic inflammation, may occur at the subclinical level in pediatric vaping patients. While the exact clinical implications of these adverse changes remain unknown, clinical symptoms supportive of the aforementioned preclinical findings may include a prolonged, wet and/or productive cough, recurrent wheezing or dyspnea in a non-asthmatic patient or recurrent throat clearing. Clinical signs may include hypoxemia, tachypnea, hoarse voice or unexplained systemic inflammation. When some or all of these supportive signs or symptoms are identified, adolescent and young adult patients should be screened for e-cigarette use including the use of the novel disposable vaping products (79). In the appropriate clinical context of recurrent respiratory infections, healthcare providers may evaluate for early signs of respiratory tract colonization contributing to these signs and symptoms with chest imaging, and microbiological workup for common bacterial and viral pathogens and basic systemic inflammatory markers [such as C-reactive protein (CRP)].
Future clinical studies are required to help validate preclinical findings of e-cigarette exposure contributing to an increased susceptibility of respiratory tract infections secondary to impaired innate immunity. More specifically, prospective studies assessing for relevant clinical outcomes of respiratory infection are needed. Examples of pertinent outcomes include recurrent lower respiratory signs and symptoms, respiratory cultures from the upper and lower airways including novel pathogens, changes on chest imaging, hospitalization rates, antibiotic use or other treatments for lower respiratory tract infections. Future clinical trials will also help to identify whether certain populations are more susceptible to respiratory infections, such as those with underlying respiratory conditions, younger or older age groups, and/or certain gender or ethnic groups. Until this occurs, the most effective measure to prevent lung disease from inhalation exposure is abstinence from e-cigarette products use. Researchers, healthcare providers, and parents should continue to advocate for more stringent e-cigarette regulations, legislative action, and counter promotion to protect children and young adults from starting to e-cigarette use. The intent of this review was to provide education to healthcare providers and researchers on the potential mechanisms of innate immune impairment secondary to e-cigarette exposure that may result in future e-cigarette policies protecting the pediatric population.
Acknowledgments
Funding: Grants from the National Institute of Environmental Health Sciences P30 ES001247 (DPC, MDM) and L40 ES030909-01 (MDM), National Heart Lung and Blood Institute 5R01HL139335 (MDM), National Center for Advancing Translational Sciences 2KL2TR001999 (MDM), and National Cancer Institute U54CA228110 (DPC, RSM, MDM) as well as the David H. Smith Fund at URMC (MDM).
Footnote
Reporting Checklist: Authors have completed the narrative review reporting checklist. Available at http://dx.doi.org/10.21037/pm-20-97
Peer Review File: Available at http://dx.doi.org/10.21037/pm-20-97
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form at the time of submission (Available at http://dx.doi.org/10.21037/pm-20-97). COI’s are equivalent to those sources of funding identified above in Sources of Funding (DPC, RSM, MDM).
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Cullen KA, Ambrose BK, Gentzke AS, et al. Notes from the Field: Use of Electronic Cigarettes and Any Tobacco Product Among Middle and High School Students - United States, 2011-2018. MMWR Morb Mortal Wkly Rep 2018;67:1276-7. [Crossref] [PubMed]
- Wang TW. E-cigarette Use Among Middle and High School Students - United States, 2020. MMWR Morb Mortal Wkly Rep 2020;69:1310-2. [Crossref] [PubMed]
- Bullen C, Howe C, Laugesen M, et al. Electronic cigarettes for smoking cessation: a randomised controlled trial. Lancet 2013;382:1629-37. [Crossref] [PubMed]
- Hartmann‐Boyce J, McRobbie H, Bullen C, et al. Electronic cigarettes for smoking cessation. Cochrane Database Syst Rev 2016;9:CD010216 [PubMed]
- Kalkhoran S, Glantz SA. E-cigarettes and smoking cessation in real-world and clinical settings: a systematic review and meta-analysis. Lancet Respir Med 2016;4:116-28. [Crossref] [PubMed]
- Raber JC, Elzinga S, Kaplan C. Understanding dabs: contamination concerns of cannabis concentrates and cannabinoid transfer during the act of dabbing. J Toxicol Sci 2015;40:797-803. [Crossref] [PubMed]
- Layden JE, Ghinai I, Pray I, et al. Pulmonary illness related to e-cigarette use in Illinois and Wisconsin - Final Report. N Engl J Med 2020;382:903-16. [Crossref] [PubMed]
- Krishnasamy VP, Hallowell BD, Ko JY, et al. Update: characteristics of a nationwide outbreak of e-cigarette, or vaping, product use–associated lung injury - United States, August 2019-January 2020. MMWR Morb Mortal Wkly Rep 2020;69:90. [Crossref] [PubMed]
- Xu JQ, Murphy SL, Kochanek KD, et al. Mortality in the United States, 2018. NCHS Data Brief, no 355. Hyattsville, MD: National Center for Health Statistics. 2020.
- CDC. Estimated Influenza Illnesses, Medical visits, Hospitalizations, and Deaths in the United States – 2019-2020 Influenza Season 2020 [cited 2020. CDC estimated influenza visits]. Available online: https://www.cdc.gov/flu/about/burden/2019-2020.html
- Hall CB, Weinberg GA, Iwane MK, et al. The burden of respiratory syncytial virus infection in young children. N Engl J Med 2009;360:588-98. [Crossref] [PubMed]
- Control CfD, Prevention. United States COVID-19 cases and deaths by state CDC: CDC; 2020 [cited 2020. CDC COVID-19 Summary]. Available online: https://www.cdc.gov/coronavirus/2019-ncov/cases-updates/cases-in-us.html
- Bridge R, Brady S, Erhart LM, et al. Notes from the Field: Age Distribution of Patients with Laboratory-Detected Respiratory Syncytial Virus - Arizona, 2013-2017. MMWR Morb Mortal Wkly Rep 2019;68:203-4. [Crossref] [PubMed]
- Boehmer TK, DeVies J, Caruso E, et al. Changing age distribution of the COVID-19 pandemic - United States, May-August 2020. MMWR Morb Mortal Wkly Rep 2020;69:1404-9. [Crossref] [PubMed]
- Heron M. Deaths: Leading causes for 2017. Natl Vital Stat Rep 2019;68:1-77. [PubMed]
- Lynch JP 3rd, Zhanel GG. Streptococcus pneumoniae: epidemiology and risk factors, evolution of antimicrobial resistance, and impact of vaccines. Curr Opin Pulm Med 2010;16:217-25. [Crossref] [PubMed]
- Soeters HM, Blain A, Pondo T, et al. Current epidemiology and trends in invasive Haemophilus influenzae disease - United States, 2009-2015. Clin Infect Dis 2018;67:881-9. [Crossref] [PubMed]
- Hahn J, Monakhova YB, Hengen J, et al. Electronic cigarettes: overview of chemical composition and exposure estimation. Tob Induc Dis 2014;12:23. [Crossref] [PubMed]
- Varlet V, Farsalinos K, Augsburger M, et al. Toxicity assessment of refill liquids for electronic cigarettes. Int J Environ Res Public Health 2015;12:4796-815. [Crossref] [PubMed]
- Duffy B, Li L, Lu S, et al. Analysis of cannabinoid-containing fluids in illicit vaping cartridges recovered from pulmonary injury patients: identification of vitamin E acetate as a major diluent. Toxics 2020;8:8. [Crossref] [PubMed]
- Bhat TA, Kalathil SG, Bogner PN, et al. An Animal Model of Inhaled Vitamin E Acetate and EVALI-like Lung Injury. N Engl J Med 2020;382:1175-7. [Crossref] [PubMed]
- Attfield KR, Chen W, Cummings KJ, et al. Potential of Ethenone (Ketene) to Contribute to Electronic Cigarette, or Vaping, Product Use-associated Lung Injury. Am J Respir Crit Care Med 2020;202:1187-9. [Crossref] [PubMed]
- Hajek P, Etter JF, Benowitz N, et al. Electronic cigarettes: review of use, content, safety, effects on smokers and potential for harm and benefit. Addiction 2014;109:1801-10. [Crossref] [PubMed]
- Cheng T. Chemical evaluation of electronic cigarettes. Tob Control 2014;23:ii11-ii17. [Crossref] [PubMed]
- Farsalinos KE, Gillman G. Carbonyl emissions in e-cigarette aerosol: a systematic review and methodological considerations. Front Physiol 2018;8:1119. [Crossref] [PubMed]
- Grana R, Benowitz N, Glantz SA. E-cigarettes: a scientific review. Circulation 2014;129:1972-86. [Crossref] [PubMed]
- Quinton LJ, Walkey AJ, Mizgerd JP. Integrative physiology of pneumonia. Physiol Rev 2018;98:1417-64. [Crossref] [PubMed]
- Bonilla FA, Oettgen HC. Adaptive immunity. J Allergy Clin Immunol 2010;125:S33-S40. [Crossref] [PubMed]
- Curtis JL. Cell-mediated adaptive immune defense of the lungs. Proc Am Thorac Soc 2005;2:412-6. [Crossref] [PubMed]
- Bagale K, Paudel S, Cagle H, et al. Electronic Cigarette (E-Cigarette) Vapor Exposure Alters the Streptococcus pneumoniae Transcriptome in a Nicotine-Dependent Manner without Affecting Pneumococcal Virulence. Appl Environ Microbiol 2020;86:e02125-19. [PubMed]
- Corriden R, Moshensky A, Bojanowski CM, et al. E-cigarette use increases susceptibility to bacterial infection by impairment of human neutrophil chemotaxis, phagocytosis, and NET formation. Am J Physiol Cell Physiol 2020;318:C205-C214. [Crossref] [PubMed]
- Hwang JH, Lyes M, Sladewski K, et al. Electronic cigarette inhalation alters innate immunity and airway cytokines while increasing the virulence of colonizing bacteria. J Mol Med (Berl) 2016;94:667-79. [Crossref] [PubMed]
- Madison MC, Landers CT, Gu BH, et al. Electronic cigarettes disrupt lung lipid homeostasis and innate immunity independent of nicotine. J Clin Invest 2019;129:4290-304. [Crossref] [PubMed]
- Sussan TE, Gajghate S, Thimmulappa RK, et al. Exposure to electronic cigarettes impairs pulmonary anti-bacterial and anti-viral defenses in a mouse model. PLoS One 2015;10:e0116861 [Crossref] [PubMed]
- Crotty Alexander LE, Drummond CA, et al. Chronic inhalation of e-cigarette vapor containing nicotine disrupts airway barrier function and induces systemic inflammation and multiorgan fibrosis in mice. Am J Physiol Regul Integr Comp Physiol 2018;314:R834-47. [Crossref] [PubMed]
- Gómez AC, Rodríguez-Fernández P, Villar-Hernández R, et al. E-cigarettes: Effects in phagocytosis and cytokines response against Mycobacterium tuberculosis. PLoS One 2020;15:e0228919 [Crossref] [PubMed]
- Scott A, Lugg ST, Aldridge K, et al. Pro-inflammatory effects of e-cigarette vapour condensate on human alveolar macrophages. Thorax 2018;73:1161-9. [Crossref] [PubMed]
- Ween MP, Whittall JJ, Hamon R, et al. Phagocytosis and Inflammation: Exploring the effects of the components of E‐cigarette vapor on macrophages. Physiol Rep 2017;5:e13370 [Crossref] [PubMed]
- Wu Q, Di Jiang MM, Chu HW. Electronic cigarette liquid increases inflammation and virus infection in primary human airway epithelial cells. PLoS One 2014;9:e108342 [Crossref] [PubMed]
- Haq IJ, Gray MA, Garnett JP, et al. Airway surface liquid homeostasis in cystic fibrosis: pathophysiology and therapeutic targets. Thorax 2016;71:284-7. [Crossref] [PubMed]
- Randell SH, Boucher RC. Effective mucus clearance is essential for respiratory health. Am J Respir Cell Mol Biol 2006;35:20-8. [Crossref] [PubMed]
- Button B, Cai LH, Ehre C, et al. A periciliary brush promotes the lung health by separating the mucus layer from airway epithelia. Science 2012;337:937-41. [Crossref] [PubMed]
- Pezzulo AA, Tang XX, Hoegger MJ, et al. Reduced airway surface pH impairs bacterial killing in the porcine cystic fibrosis lung. Nature 2012;487:109-13. [Crossref] [PubMed]
- Boucher RC. Airway surface dehydration in cystic fibrosis: pathogenesis and therapy. Annu Rev Med 2007;58:157-70. [Crossref] [PubMed]
- Chung S, Baumlin N, Dennis JS, et al. Electronic cigarette vapor with nicotine causes airway mucociliary dysfunction preferentially via TRPA1 receptors. Am J Respir Crit Care Med 2019;200:1134-45. [Crossref] [PubMed]
- Lin VY, Fain MD, Jackson PL, et al. Vaporized e-cigarette liquids induce ion transport dysfunction in airway epithelia. Am J Respir Cell Mol Biol 2019;61:162-73. [Crossref] [PubMed]
- Sherwood CL, Boitano S. Airway epithelial cell exposure to distinct e-cigarette liquid flavorings reveals toxicity thresholds and activation of CFTR by the chocolate flavoring 2, 5-dimethypyrazine. Respir Res 2016;17:57. [Crossref] [PubMed]
- Rayner RE, Makena P, Prasad GL, et al. Cigarette and ENDS preparations differentially regulate ion channels and mucociliary clearance in primary normal human bronchial 3D cultures. Am J Physiol Lung Cell Mol Physiol 2019;317:L295-L302. [Crossref] [PubMed]
- Sosnowski TR, Jabłczyńska K, Odziomek M, et al. Physicochemical studies of direct interactions between lung surfactant and components of electronic cigarettes liquid mixtures. Inhal Toxicol 2018;30:159-68. [Crossref] [PubMed]
- Przybyla RJ, Wright J, Parthiban R, et al. Electronic cigarette vapor alters the lateral structure but not tensiometric properties of calf lung surfactant. Respir Res 2017;18:193. [Crossref] [PubMed]
- Blount BC, Karwowski MP, Shields PG, et al. Vitamin E acetate in bronchoalveolar-lavage fluid associated with EVALI. N Engl J Med 2020;382:697-705. [Crossref] [PubMed]
- DiPasquale M, Gbadamosi O, Nguyen MH, et al. A Mechanical Mechanism for Vitamin E Acetate in E-cigarette/Vaping-Associated Lung Injury. Chem Res Toxicol 2020;33:2432-40. [Crossref] [PubMed]
- Lachowicz-Scroggins ME, Yuan S, Kerr SC, et al. Abnormalities in MUC5AC and MUC5B protein in airway mucus in asthma. Am J Respir Crit Care Med 2016;194:1296-9. [Crossref] [PubMed]
- Gellatly S, Pavelka N, Crue T, et al. Nicotine-Free e-Cigarette Vapor Exposure Stimulates IL6 and Mucin Production in Human Primary Small Airway Epithelial Cells. J Inflamm Res 2020;13:175-85. [Crossref] [PubMed]
- Neveu WA, Allard JB, Dienz O, et al. IL-6 is required for airway mucus production induced by inhaled fungal allergens. J Immunol 2009;183:1732-8. [Crossref] [PubMed]
- Mercer RR, Russell ML, Roggli VL, et al. Cell number and distribution in human and rat airways. Am J Respir Cell Mol Biol 1994;10:613-24. [Crossref] [PubMed]
- Knight DA, Holgate ST. The airway epithelium: structural and functional properties in health and disease. Respirology 2003;8:432-46. [Crossref] [PubMed]
- Crystal RG, Randell SH, Engelhardt JF, et al. Airway epithelial cells: current concepts and challenges. Proc Am Thorac Soc 2008;5:772-7. [Crossref] [PubMed]
- Adams M, Smith UM, Logan CV, et al. Recent advances in the molecular pathology, cell biology and genetics of ciliopathies. J Med Genet 2008;45:257-67. [Crossref] [PubMed]
- Eisele NA, Anderson DM. Host defense and the airway epithelium: frontline responses that protect against bacterial invasion and pneumonia. J Pathog 2011;2011:249802 [Crossref] [PubMed]
- Hiemstra PS, McCray PB, Bals R. The innate immune function of airway epithelial cells in inflammatory lung disease. Eur Respir J 2015;45:1150-62. [Crossref] [PubMed]
- Vareille M, Kieninger E, Edwards MR, et al. The airway epithelium: soldier in the fight against respiratory viruses. Clin Microbiol Rev 2011;24:210-29. [Crossref] [PubMed]
- Parker D, Prince A. Innate immunity in the respiratory epithelium. Am J Respir Cell Mol Biol 2011;45:189-201. [Crossref] [PubMed]
- Johansson N, Kalin M, Tiveljung-Lindell A, et al. Etiology of community-acquired pneumonia: increased microbiological yield with new diagnostic methods. Clin Infect Dis 2010;50:202-9. [Crossref] [PubMed]
- Hergott CB, Roche AM, Naidu NA, et al. Bacterial exploitation of phosphorylcholine mimicry suppresses inflammation to promote airway infection. J Clin Invest 2015;125:3878-90. [Crossref] [PubMed]
- Bogaert D, de Groot R, Hermans P. Streptococcus pneumoniae colonisation: the key to pneumococcal disease. Lancet Infect Dis 2004;4:144-54. [Crossref] [PubMed]
- Sakr A, Brégeon F, Mège JL, et al. Staphylococcus aureus nasal colonization: an update on mechanisms, epidemiology, risk factors, and subsequent infections. Front Microbiol 2018;9:2419. [Crossref] [PubMed]
- Stock I. Human rhinovirus diseases--epidemiology, treatment and prevention. Med Monatsschr Pharm 2014;37:44-53. [PubMed]
- Gally F, Di YP, Smith SK, et al. SPLUNC1 promotes lung innate defense against Mycoplasma pneumoniae infection in mice. Am J Pathol 2011;178:2159-67. [Crossref] [PubMed]
- Liegeois M, Legrand C, Desmet CJ, et al. The interstitial macrophage: A long-neglected piece in the puzzle of lung immunity. Cell Immunol 2018;330:91-6. [Crossref] [PubMed]
- Kulikauskaite J, Wack A. Teaching Old Dogs New Tricks? The Plasticity of Lung Alveolar Macrophage Subsets. Trends Immunol 2020;41:864-77. [Crossref] [PubMed]
- Ma Q. Polarization of immune cells in the pathologic response to inhaled particulates. Front Immunol 2020;11:1060. [Crossref] [PubMed]
- Jubrail J, Kurian N, Niedergang F. Macrophage phagocytosis cracking the defect code in COPD. Biomed J 2017;40:305-12. [Crossref] [PubMed]
- Miles PR, Ma J, Bowman L. Degradation of pulmonary surfactant disaturated phosphatidylcholines by alveolar macrophages. J Appl Physiol 1988;64:2474-81. [Crossref] [PubMed]
- Clapp PW, Pawlak EA, Lackey JT, et al. Flavored e-cigarette liquids and cinnamaldehyde impair respiratory innate immune cell function. Am J Physiol Lung Cell Mol Physiol 2017;313:L278-92. [Crossref] [PubMed]
- Chroneos ZC, Sever-Chroneos Z, Shepherd V. Pulmonary surfactant: an immunological perspective. Cell Physiol Biochem 2010;25:13-26. [Crossref] [PubMed]
- Tecchio C, Cassatella MA. Neutrophil-derived chemokines on the road to immunity. Semin Immunol 2016;28:119-28. [Crossref] [PubMed]
- Reidel B, Radicioni G, Clapp PW, et al. E-cigarette use causes a unique innate immune response in the lung, involving increased neutrophilic activation and altered mucin secretion. Am J Respir Crit Care Med 2018;197:492-501. [Crossref] [PubMed]
- Jenssen BP, Walley SC. Section on Tobacco Control. E-Cigarettes and Similar Devices. Pediatrics 2019;143:e20183652 [Crossref] [PubMed]
Cite this article as: Kalininskiy A, Kittel J, Nacca NE, Misra RS, Croft DP, McGraw MD. E-cigarette exposures, respiratory tract infections, and impaired innate immunity: a narrative review. Pediatr Med 2021;4:5.

