Multidisciplinary team approach for pediatric thoracic tumors: new horizons and oldies but goldies
Each year approximately 16,400 children and adolescents younger than 20 years are diagnosed with cancer in the United States (12,000 cases among children 0 to 14 years and 4,400 cases among 15 to 19 years old), with an incidence of 15.8 cases per 100,000 person-years for children and 21.7 cases per 100,000 person-years for adolescents (1).
Childhood cancer remains the leading cause of disease-related mortality among children 1 to 14 years of age, and there were approximately 1,300 cancer-related death in 2010 in the United States among children younger than 15 years, while the overall mortality for 15- to 19-year-old is lower and approximately accounts 600 deaths from cancer occurred in 2010 in this age group.
Children and adolescents tumors are rare disease, with a peculiar biology and pathogenesis, that make them very different from adult’s cancer.
In particular pediatric thoracic tumors account for 15–20% of all malignant tumors in childhood.
Most primary thoracic tumors arise in the mediastinum and their location can lead to diagnosis. Indeed, masses of the anterior mediastinum usually include lymphomas, thymic masses, teratomas, thyroid tumors. Hodgkin lymphoma is common in the middle mediastinum, whereas posterior mediastinal tumors are often neurogenic, ranging from neuroblastoma to ganglioneuroblastoma to benign tumors (2).
Primary lung tumors in children are rare, with a narrow range of diagnostic considerations. However, the overlapping imaging appearances of these tumors necessitate attention to identifying key discriminating imaging and pathologic features. In the neonate and infant, the important considerations include pleuropulmonary blastoma (PPB), infantile fibrosarcoma, and fetal lung interstitial tumor. Among these tumors, imaging findings such as air-filled cysts in type 1 PPB and homogeneously low attenuation of fetal lung interstitial tumors are relatively specific. Key pathologic and genetic discriminators among this group of tumors include the DICER1 germline mutation found in PPB and the t(12,15)(p13;q25) translocation and ETV6-NTRK3 fusion gene seen in infantile fibrosarcoma. Other rare tumors originate from the lung such as rhabdomyosarcomas, synovial sarcoma, inflammatory myofibroblastic tumors, carcinoid salivary gland-type tumors of the lung, recurrent respiratory papillomatosis, and other rare entities. Moreover, there are other malignant neoplasms that may occur in the thoracic cavity: for example, Ewing sarcoma family tumors can arise from a rib or out of the thoracic cavity, in case of extraosseous Ewing sarcoma, other types of soft-tissue sarcomas, and Langerhans cell histiocytosis (which can develop in a rib, but is not a neoplasm).
Apart from primary tumors, metastases may be located within the thorax. Nodal metastases from tumors such as neuroblastoma, rhabdomyosarcoma and germ cell tumors may occur in the middle mediastinum. Pulmonary metastases usually derive from osteosarcoma, Ewing sarcoma, Wilms tumor, hepatoblastoma, germ cell tumors, rhabdomyosarcoma, synovial sarcomas and other soft-tissue sarcomas (3). Eventually, tumors like osteosarcoma, neuroblastoma, Ewing sarcoma and rhabdomyosarcoma can determine bone metastases affecting the thoracic wall.
The challenge in diagnosing pediatric cancers is that they occur so infrequently and often initially present with symptoms compatible with more common and less serious illnesses. Adding to these early detection difficulties is the fact that once a specific cancer has been diagnosed in a practice, not only will there be a long lag time until the next case presents, but the next case is likely to be an entirely different entity with different presenting features.
In spite of the inherent difficulties in diagnosis, timely diagnosis of childhood cancer is extremely important. Many pediatric malignancies are highly curable, and with some of them, earlier diagnosis can be associated with a better prognosis, diminished intensity of therapy, and less complications from disease as well as treatment.
If a diagnosis of a cancer is suspected or confirmed, the patient must be immediately referred to a pediatric cancer center. Given the rarity, different types of cancer, different chemotherapy and radiotherapy sensibility, and peculiar surgery approach, is crucial the management of these rare disease by the dedicated and specific specialists.
This means that children and families often need to travel great distances to receive state-of-art care, frequently with considerable costs and enormous strains on family and other social anchors that are so essential for normal children development.
The multimodal approach, which integrates surgery and radiotherapy to control local disease with chemotherapy to eradicate systemic (metastatic) disease, has become the standard approach to treating most childhood cancers. Nowadays the 5-year survival rate for all forms of childhood cancer is 83%. The striking improvement in survival is a direct result of incorporation of anticancer drugs into treatment regimens that previously relied only on surgery or radiotherapy for primary tumor. Ongoing advances in our understanding of the biology of pediatric cancers, that will allow to define the role of an expanding number of agents that target cell-signalling pathways that contribute to but are not causative of the malignant process, will be the next therapeutic challenge for pediatric cancer drug development (4). Greater resources and efforts will be required to meet these challenges and will required improved integration of advances in the biological and pharmacological basic science into the design and use of chemotherapeutic regimens.
Surgery remains a critical component in the multimodal therapy of childhood cancer. Accurate staging and successful resection of a tumor is of vital importance as we strive to minimize the morbidity of treatment. Overall the past 15 years, minimally invasive surgery procedures have become common practice. Improved optics, new trocar systems, and specifically designed instruments now allow a wide spectrum of surgical operations to be performed without the use of large thoracotomies. Oncological surgical principles, such as adequate exploration, complete excision of the mass with an appropriate margin, and minimizing the risk of a tumor spill, are still paramount. Thoracoscopic surgery is usually performed under general anaesthesia and permits to remove nodules located peripherally in then lung, along the parietal pleura, or on the diaphragm. Anyway, osteogenic sarcoma metastatic to the lung should be managed by standard thoracotomy, that permit palpation of small intraparenchymal sarcoma metastases. The mediastinum is also accessible by minimally invasive techniques for thoracoscopically guided needle biopsies, excisional or incisional biopsies, as well as for tumor removal in selected individuals.
As a discipline, pediatric oncology also stands as a paradigm for cancer research. The majority of children diagnosed with cancer are enrolled in cooperative clinical trials that provide state-of-the-art therapy at the centres across the world. This strong collaboration and cooperation, that is the milestone of the pediatric cancer community, and has allowed the creation of networks fundamental for translational research discoveries and new investigators (5).
Discoveries and insights emanating from the rapidly evolving fields of cancer biology, immunology, genetics, and clinical pharmacology, has meant that basic science innovations were rapidly translated into clinical trials contributing to one of the most remarkable achievement in medicine. In less than 50 years, many children cancers have advanced from incurable to curable and others have become treatable. So pediatric oncology is now a discipline that offers hope along with challenge.
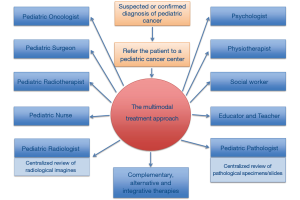
Despite this progress, the care of children requires the collaborations of many disciplines and individuals: pediatric oncologists, radiation therapists, surgeons, radiologists, and pathologists work collaboratively with nurses, physician’s assistants, social workers, nutritionists, psychologists, physiotherapists, pharmacists, educators, teachers, and of course, parents and families (Figure 1). The collaboration is crucial and essential to treating the patients in the best way (6).
Usually each single case of pediatric tumor is discussed with all specialists in a multidisciplinary meeting in which the radiological imagine and pathological specimens are revised; moreover, pathological samples are usually revised in a setting of centralized pathological anatomy review area (7). Sometimes pathological slides are revised and discussed during conference call. The same thing is possible for discussing and reviewing radiological imagines and for sharing particular cases with other pediatric oncologists that are located in different hospitals. The modern technologies permit to send very quickly radiological examinations, for example via File Transfer Protocol (FTP), but sometimes CD-rooms are sent from one hospital to another hospital to be revised and discussed. In so rare disease the concept of “oncological network or rare disease network” is pregnant, and every effort must be done to share and discuss with different expert specialists. So we are firmly convinced that new technologies and telemedicine can be very useful in such rare pathologies, but we are also resolute that only expert pediatric oncologists must take care of this setting of patients.
Moreover, we stress the importance of the support of the other different specialists as physiotherapists, psychologists, nurses, social workers, educators, teachers, because of each patient should be able to continue to live his life as far as possible, continuing the school path, with the possibility to also carry out schooling at home and continue sport activity, if possible also during the hospitalization, taking into account the existence of gyms in some hospitals.
Since families and patients are often forced to move from home to receive state-of-art care, for a long time, is important to consider costs and enormous strains on family and other social anchors, and support families financially and morally, helping them to find hospitalities houses. New technologies help families also in this area, making travelling easier.
We take in mind that cooperation is basilar and that Pediatric Cancer Unit does not exist in isolation for the delivery of treatment: it should be an element of regional and national networks that joint forces to address the travel, lodging, food and financial needs of families who must leave their homes for prolonged periods. These needs require that the health care professionals establish long-term collaboration with administrators and policy makers, and they seek partnership with community groups, including parent/patient support, advocacy, and fund-raising group (8,9).
An emerging discussion point is represented by complementary, alternative and integrative oncology therapies. What does it mean? It is the set of those therapies and activities that are provided to better take care for patients, in addition to their conventional cancer treatment, and focusing on wellness and health promotion, such as yoga, shiatsu, massage, guided imagery, biofeedback, mindfulness, acupuncture, dietary supplements, as other therapies that work more on the mind level such as hypnosis and relaxation techniques. Holistic medicine refers to the care of patient’s body, emotions, spirit, and relationship, as a whole, in the context of the patient’s values, beliefs, family, culture, and community. Adding to this complexity is the growing attention to the notion of integrated care programs in oncology, in which numerous cancer specialties work together to provide comprehensive patient care.
Treatment of pediatric cancer is expensive; because of the rarity of childhood cancer and its small impact on the overall under-5 mortality rates, many “low-to-middle income countries” (LMIC) have not budgeted for the treatment of children with cancer (10). The International Society of Pediatric Oncology (SIOP) and the International Confederation of Childhood Cancer Parent’s Organization (ICCPO) have collaborated in the development of policy issues to improve access to care. By using recent delivery options, including international partnerships and twinning with the aid of information and communication technology and telemedicine, the cancer divisory between high-income countries and LMIC can be bridged and the barriers to effective cancer care and control can be overcome. The sharing of experience, commitment, and resources is the basis of the successful “twinning” programs that have been established between large pediatric oncology centres in developed countries and aspiring Institutions in LMIC.
Pediatric cancers confront the health care system with a formidable challenge. Cancer is a particularly dreaded disease, and children among the most vulnerable members of society. Moreover, preventing childhood cancer deaths can extend lives by many decades. Nonetheless, addressing cancer is expensive because it involves the use of sophisticated diagnostic tools and new medications. Rising health care costs associated with new technologies threaten the financial viability of a health care system already struggling to deal with the growing number of older population: this must be a reflection point.
Acknowledgments
The authors would like to acknowledge Associazione Bianca Garavaglia Onlus.
Funding: None.
Footnote
Provenance and Peer Review: This article was commissioned by the Guest Editors (Paolo Scanagatta) for the Series “Pediatric Thoracic Surgery” published in Pediatric Medicine. The article has undergone external peer review.
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/pm.2019.04.02). The series “Pediatric Thoracic Surgery” was commissioned by the editorial office without any funding or sponsorship. The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Linabery AM, Ross JA. Trends in childhood cancer incidence in the U.S. (1992-2004). Cancer 2008;112:416-32. [Crossref] [PubMed]
- Lichtenberger JP 3rd, Biko DM, Carter BW, et al. Primary Lung Tumors in Children: Radiologic-Pathologic Correlation From the Radiologic Pathology Archives. Radiographics 2018;38:2151-72. [Crossref] [PubMed]
- Tronc F, Conter C, Marec-Berard P, et al. Prognostic factors and long-term results of pulmonary metastasectomy for pediatric histologies. Eur J Cardiothorac Surg 2008;34:1240-6. [Crossref] [PubMed]
- Filbin M, Monje M. Developmental origins and emerging therapeutic opportunities for childhood cancer. Nat Med 2019;25:367-76. [Crossref] [PubMed]
- Witt CM, Balneaves LG, Cardoso MJ, et al. A Comprehensive Definition for Integrative Oncology. J Natl Cancer Inst Monogr 2017; [Crossref] [PubMed]
- Eiser C, Hill JJ, Vance YH. Examining the psychological consequences of surviving childhood cancer: systematic review as a research method in pediatric psychology. J Pediatr Psychol 2000;25:449-60. [Crossref] [PubMed]
- Ouwens M, Hulscher M, Hermens R, et al. Implementation of integrated care for patients with cancer: a systematic review of interventions and effects. Int J Qual Health Care 2009;21:137-44. [Crossref] [PubMed]
- Jacobs S, Mowbray C. The Power of Massage in Children with Cancer-How Can We Do Effective Research? Children (Basel) 2019; [Crossref] [PubMed]
- Seelisch J, Sung L, Kelly MJ, et al. Identifying clinical practice guidelines for the supportive care of children with cancer: A report from the Children's Oncology Group. Pediatr Blood Cancer 2019;66:e27471 [Crossref] [PubMed]
- Cameron A, Ewen M, Ross-Degnan D, et al. Medicine prices, availability, and affordability in 36 developing and middle-income countries: a secondary analysis. Lancet 2009;373:240-9. [Crossref] [PubMed]
Cite this article as: Meazza C, Gattuso G. Multidisciplinary team approach for pediatric thoracic tumors: new horizons and oldies but goldies. Pediatr Med 2019;2:14.